|
|
-
We minimize duplication of effort by augmenting your current IT infrastructure
-
We have a rigorous automated review and audit process to minimize problem resolution time.
-
We have an efficient process flow to resolve issues that minimize the impact on your registry or provider's organization
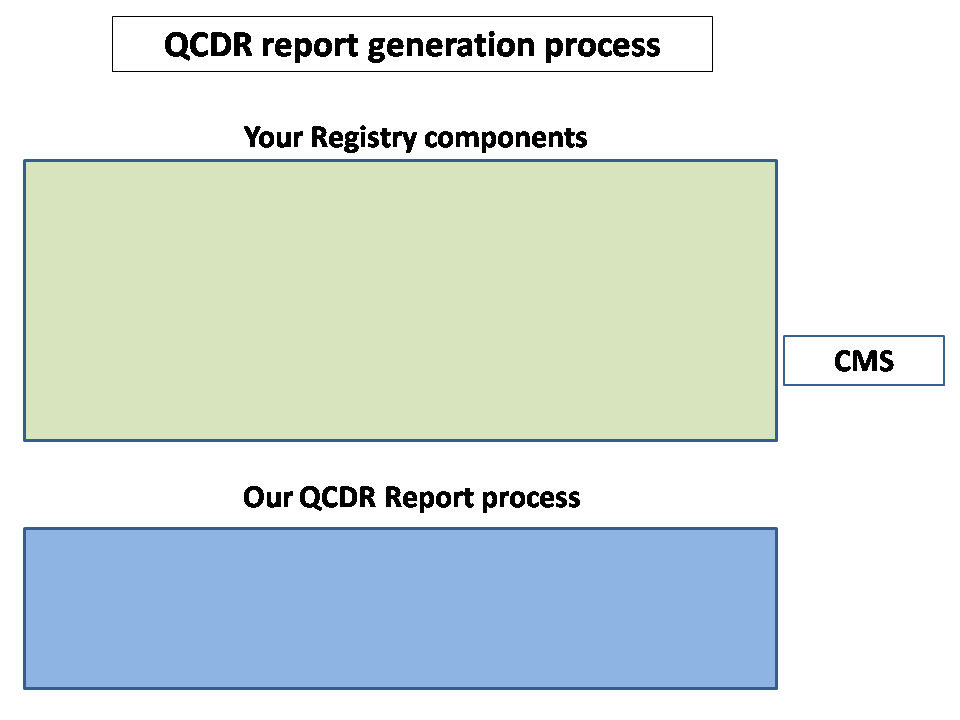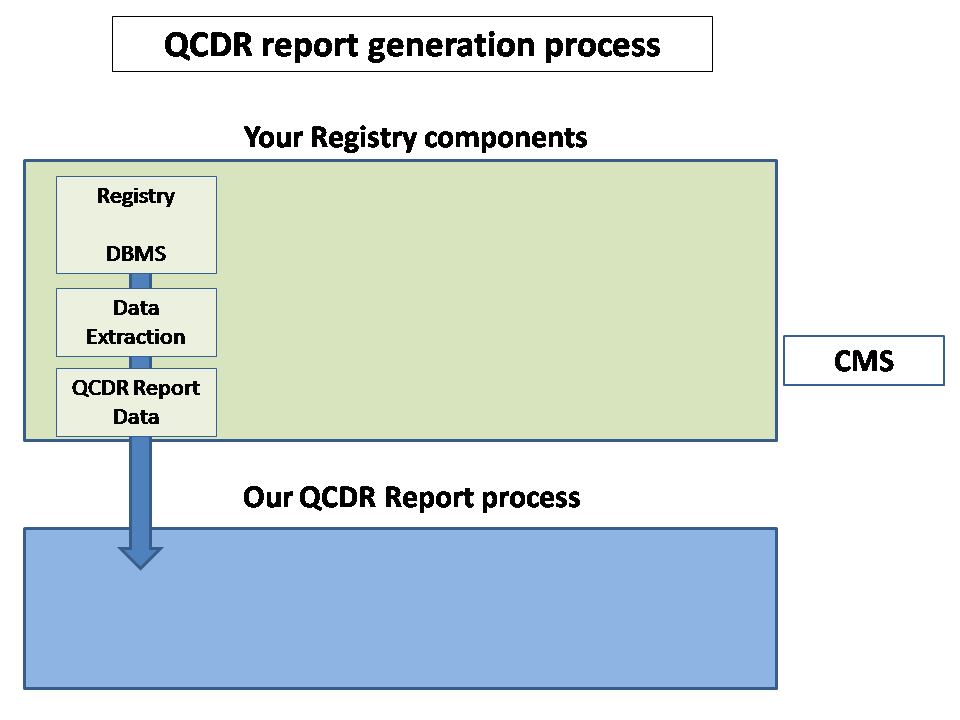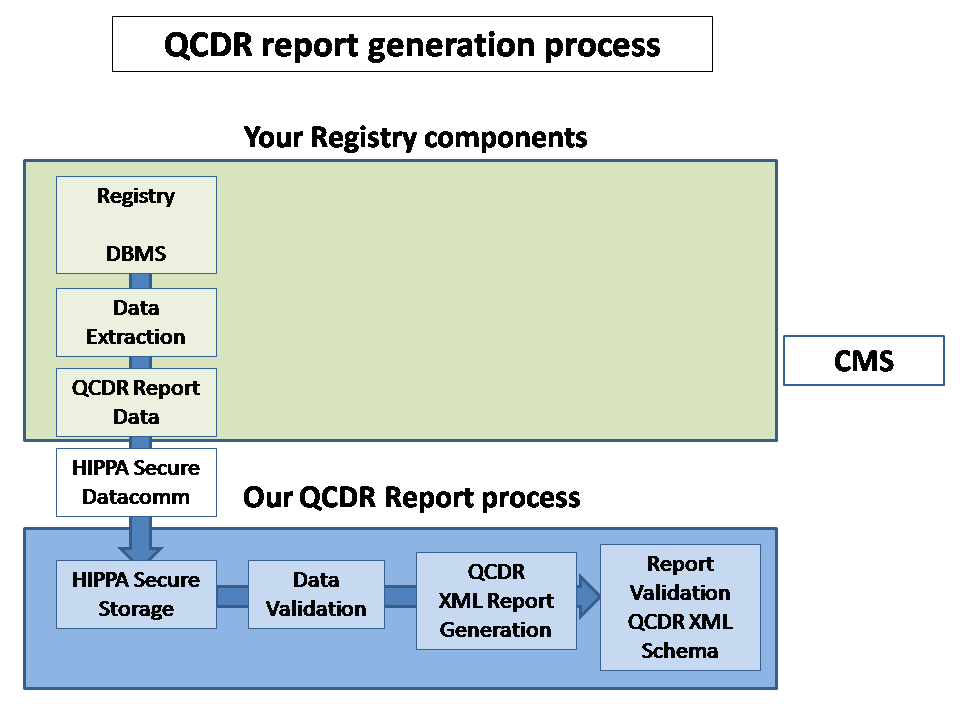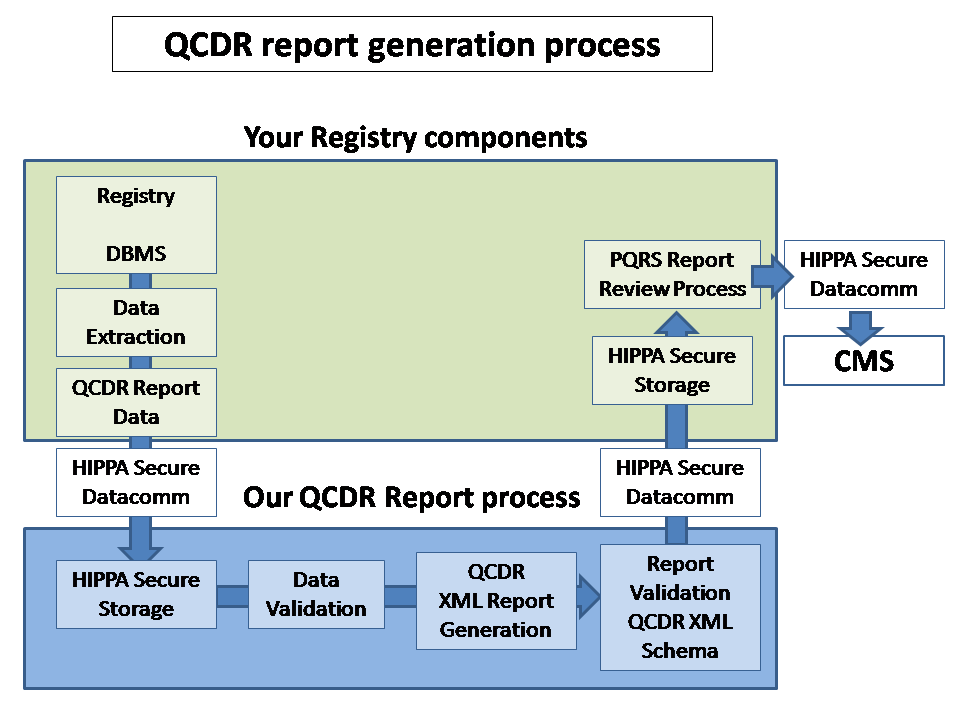
In order
to fulfill CMS requirements for Qualified Clinical Data Registry (QCDR),
CUHSM.ORG
compiles and validates CMS submissions with our Submission Generation Engine
by using our efficient Data-Mining processes for
patient experience databases and
provider EHRs.